National
Year in review: Home HIV tests become available
Makers hope new anonymous home testing option will increase testing rates
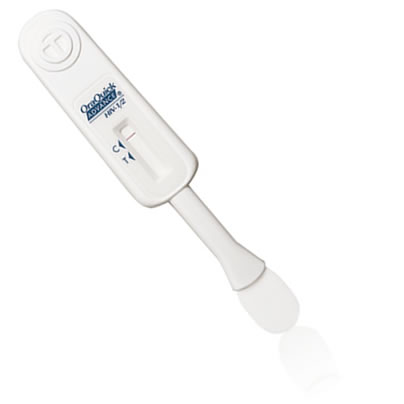
Known as the OraQuick In-Home HIV test, the test was developed by OraSure Technologies, Inc.
“The test has the potential to identify large numbers of previously undiagnosed HIV infections, especially if used by those unlikely to use standard screening methods,” the FDA said in a new release.
“Knowing your status is an important factor in the effort to prevent the spread of HIV,” said Dr. Karen Midthun, director of the FDA’s Center for Biologics Evaluation and Research. “The availability of a home-use HIV test kit provides another option for individuals to get tested so that they can seek medical care, if appropriate.”
The FDA says clinical studies of the test showed a 92 percent sensitivity rate, which means that of every 12 HIV-infected individuals tested with the kit, one negative result could be expected.
“A positive result with this test does not mean that an individual is definitely infected with HIV, but rather that additional testing should be done in a medical setting to confirm the result,” the FDA said. “Similarly, a negative test result does not mean that an individual is definitely not infected with HIV, particularly when exposure may have been within the previous three months.”
FDA officials noted that the OraSure in-home test is the first HIV test that allows users to learn their results at home immediately without interacting with a lab or medical professionals.
The officials said the new test’s potential for identifying large numbers of people who don’t know they are infected outweighs concern by some that people who test positive should have immediate access to counseling and medical advice.
Experts estimate that one-fifth of the people infected with HIV are unaware of their status and often contribute to the infection of others.
U.S. Supreme Court
Supreme Court to consider bans on trans athletes in school sports
27 states have passed laws limiting participation in athletics programs

The U.S. Supreme Court on Thursday agreed to hear two cases involving transgender youth challenging bans prohibiting them from participating in school sports.
In Little v. Hecox, plaintiffs represented by the ACLU, Legal Voice, and the law firm Cooley are challenging Idaho’s 2020 ban, which requires sex testing to adjudicate questions of an athlete’s eligibility.
The 9th U.S. Circuit Court of Appeals described the process in a 2023 decision halting the policy’s enforcement pending an outcome in the litigation. The “sex dispute verification process, whereby any individual can ‘dispute’ the sex of any female student athlete in the state of Idaho,” the court wrote, would “require her to undergo intrusive medical procedures to verify her sex, including gynecological exams.”
In West Virginia v. B.P.J., Lambda Legal, the ACLU, the ACLU of West Virginia, and Cooley are representing a trans middle school student challenging the Mountain State’s 2021 ban on trans athletes.
The plaintiff was participating in cross country when the law was passed, taking puberty blockers that would have significantly reduced the chances that she could have a physiological advantage over cisgender peers.
“Like any other educational program, school athletic programs should be accessible for everyone regardless of their sex or transgender status,” said Joshua Block, senior counsel for the ACLU’s LGBTQ and HIV Project. “Trans kids play sports for the same reasons their peers do — to learn perseverance, dedication, teamwork, and to simply have fun with their friends,” Block said.
He added, “Categorically excluding kids from school sports just because they are transgender will only make our schools less safe and more hurtful places for all youth. We believe the lower courts were right to block these discriminatory laws, and we will continue to defend the freedom of all kids to play.”
“Our client just wants to play sports with her friends and peers,” said Lambda Legal Senior Counsel Tara Borelli. “Everyone understands the value of participating in team athletics, for fitness, leadership, socialization, and myriad other benefits.”
Borelli continued, “The U.S. Court of Appeals for the Fourth Circuit last April issued a thoughtful and thorough ruling allowing B.P.J. to continue participating in track events. That well-reasoned decision should stand the test of time, and we stand ready to defend it.”
Shortly after taking control of both legislative chambers, Republican members of Congress tried — unsuccessfully — to pass a national ban like those now enforced in 27 states since 2020.
Federal Government
UPenn erases Lia Thomas’s records as part of settlement with White House
University agreed to ban trans women from women’s sports teams

In a settlement with the Trump-Vance administration announced on Tuesday, the University of Pennsylvania will ban transgender athletes from competing and erase swimming records set by transgender former student Lia Thomas.
The U.S. Department of Education’s Office for Civil Rights found the university in violation of Title IX, the federal rights law barring sex based discrimination in educational institutions, by “permitting males to compete in women’s intercollegiate athletics and to occupy women-only intimate facilities.”
The statement issued by University of Pennsylvania President J. Larry Jameson highlighted how the law’s interpretation was changed substantially under President Donald Trump’s second term.
“The Department of Education OCR investigated the participation of one transgender athlete on the women’s swimming team three years ago, during the 2021-2022 swim season,” he wrote. “At that time, Penn was in compliance with NCAA eligibility rules and Title IX as then interpreted.”
Jameson continued, “Penn has always followed — and continues to follow — Title IX and the applicable policy of the NCAA regarding transgender athletes. NCAA eligibility rules changed in February 2025 with Executive Orders 14168 and 14201 and Penn will continue to adhere to these new rules.”
Writing that “we acknowledge that some student-athletes were disadvantaged by these rules” in place while Thomas was allowed to compete, the university president added, “We recognize this and will apologize to those who experienced a competitive disadvantage or experienced anxiety because of the policies in effect at the time.”
“Today’s resolution agreement with UPenn is yet another example of the Trump effect in action,” Education Secretary Linda McMahon said in a statement. “Thanks to the leadership of President Trump, UPenn has agreed both to apologize for its past Title IX violations and to ensure that women’s sports are protected at the university for future generations of female athletes.”
Under former President Joe Biden, the department’s Office of Civil Rights sought to protect against anti-LGBTQ discrimination in education, bringing investigations and enforcement actions in cases where school officials might, for example, require trans students to use restrooms and facilities consistent with their birth sex or fail to respond to peer harassment over their gender identity.
Much of the legal reasoning behind the Biden-Harris administration’s positions extended from the 2020 U.S. Supreme Court case Bostock v. Clayton County, which found that sex-based discrimination includes that which is based on sexual orientation or gender identity under Title VII rules covering employment practices.
The Trump-Vance administration last week put the state of California on notice that its trans athlete policies were, or once were, in violation of Title IX, which comes amid the ongoing battle with Maine over the same issue.
New York
Two teens shot steps from Stonewall Inn after NYC Pride parade
One of the victims remains in critical condition

On Sunday night, following the annual NYC Pride March, two girls were shot in Sheridan Square, feet away from the historic Stonewall Inn.
According to an NYPD report, the two girls, aged 16 and 17, were shot around 10:15 p.m. as Pride festivities began to wind down. The 16-year-old was struck in the head and, according to police sources, is said to be in critical condition, while the 17-year-old was said to be in stable condition.
The Washington Blade confirmed with the NYPD the details from the police reports and learned no arrests had been made as of noon Monday.
The shooting took place in the Greenwich Village neighborhood of Manhattan, mere feet away from the most famous gay bar in the city — if not the world — the Stonewall Inn. Earlier that day, hundreds of thousands of people marched down Christopher Street to celebrate 55 years of LGBTQ people standing up for their rights.
In June 1969, after police raided the Stonewall Inn, members of the LGBTQ community pushed back, sparking what became known as the Stonewall riots. Over the course of two days, LGBTQ New Yorkers protested the discriminatory policing of queer spaces across the city and mobilized to speak out — and throw bottles if need be — at officers attempting to suppress their existence.
The following year, LGBTQ people returned to the Stonewall Inn and marched through the same streets where queer New Yorkers had been arrested, marking the first “Gay Pride March” in history and declaring that LGBTQ people were not going anywhere.
New York State Assemblywoman Deborah Glick, whose district includes Greenwich Village, took to social media to comment on the shooting.
“After decades of peaceful Pride celebrations — this year gun fire and two people shot near the Stonewall Inn is a reminder that gun violence is everywhere,” the lesbian lawmaker said on X. “Guns are a problem despite the NRA BS.”
-

 U.S. Supreme Court3 days ago
U.S. Supreme Court3 days agoSupreme Court to consider bans on trans athletes in school sports
-

 Out & About3 days ago
Out & About3 days agoCelebrate the Fourth of July the gay way!
-

 Virginia3 days ago
Virginia3 days agoVa. court allows conversion therapy despite law banning it
-

 Federal Government5 days ago
Federal Government5 days agoUPenn erases Lia Thomas’s records as part of settlement with White House