Health
PrEP gets green light in Wales
Cost concerns voiced for public health care agency


PrEP has been approved for a three-year trial in Wales.
LONDON — Despite opposition, PrEP (aka Truvada), an HIV-prevention drug popular with gay men in the U.S., has been approved for a three-year trial in Wales slated to begin this summer, the BBC reports.
But the All Wales Medicines Strategy Group spoke of “several uncertanties and limitations” in the economic model presented by the drug company, the BBC reports.
The Terrence Higgins Trust in Wales called the Strategy Group’s recommendation “short sighted” and said PrEP could cost NHS Wales, the country’s publicly funded health care system, money in the long run, the BBC article said.
Around 150 new people are diagnosed with HIV in Wales each year, nearly half from sexually transmitted infection between men.
A daily dose of the pill is suggested for people at high risk who might not have safe sex. It is currently used in the US, Canada, Australia and France, and taken daily, it has been shown to reduce the risk of infection by 86 percent.
Stonewall Cymru, an LGBT charity, has been campaigning for PrEP to be made available, the BBC reports.
Monkeypox
US contributes more than $90 million to fight mpox outbreak in Africa
WHO and Africa CDC has declared a public health emergency
The U.S. has contributed more than $90 million to the fight against the mpox outbreak in Africa.
The U.S. Agency for International Development on Tuesday in a press release announced “up to an additional” $35 million “in emergency health assistance to bolster response efforts for the clade I mpox outbreak in Central and Eastern Africa, pending congressional notification.” The press release notes the Biden-Harris administration previously pledged more than $55 million to fight the outbreak in Congo and other African countries.
“The additional assistance announced today will enable USAID to continue working closely with affected countries, as well as regional and global health partners, to expand support and reduce the impact of this outbreak as it continues to evolve,” it reads. “USAID support includes assistance with surveillance, diagnostics, risk communication and community engagement, infection prevention and control, case management, and vaccination planning and coordination.”
The World Health Organization and the Africa Centers for Disease Control and Prevention last week declared the outbreak a public health emergency.
The Washington Blade last week reported there are more than 17,000 suspected mpox cases across in Congo, Uganda, Kenya, Rwanda, and other African countries. The outbreak has claimed more than 500 lives, mostly in Congo.
Health
Mpox outbreak in Africa declared global health emergency
ONE: 10 million vaccine doses needed on the continent

Medical facilities that provide treatment to gay and bisexual men in some East African countries are already collaborating with them to prevent the spread of a new wave of mpox cases after the World Health Organization on Wednesday declared a global health emergency.
The collaboration, both in Uganda and Kenya, comes amid WHO’s latest report released on Aug. 12, which reveals that nine out of every 10 reported mpox cases are men with sex as the most common cause of infection.
The global mpox outbreak report — based on data that national authorities collected between January 2022 and June of this year — notes 87,189 of the 90,410 reported cases were men. Ninety-six percent of whom were infected through sex.
Sexual contact as the leading mode of transmission accounted for 19,102 of 22,802 cases, followed by non-sexual person-to-person contact. Genital rash was the most common symptom, followed by fever and systemic rash.
The WHO report states the pattern of mpox virus transmission has persisted over the last six months, with 97 percent of new cases reporting sexual contact through oral, vaginal, or anal sex with infected people.
“Sexual transmission has been recorded in the Democratic Republic of Congo among sex workers and men who have sex with men,” the report reads. “Among cases exposed through sexual contact in the Democratic Republic of the Congo, some individuals present only with genital lesions, rather than the more typical extensive rash associated with the virus.”
The growing mpox cases, which are now more than 2,800 reported cases in at least 13 African countries that include Kenya, Uganda, Rwanda, and prompted the Africa Centers for Disease Control and Prevention this week to declare the disease a public health emergency for resource mobilization on the continent to tackle it.
“Africa has long been on the frontlines in the fight against infectious diseases, often with limited resources,” said Africa CDC Director General Jean Kaseya. “The battle against Mpox demands a global response. We need your support, expertise, and solidarity. The world cannot afford to turn a blind eye to this crisis.”
The disease has so far claimed more than 500 lives, mostly in Congo, even as the Africa CDC notes suspected mpox cases across the continent have surged past 17,000, compared to 7,146 cases in 2022 and 14,957 cases last year.
“This is just the tip of the iceberg when we consider the many weaknesses in surveillance, laboratory testing, and contact tracing,” Kaseya said.
WHO, led by Director General Tedros Adhanom Ghebreyesus, also followed the Africa CDC’s move by declaring the mpox outbreak a public health emergency of international concern.
The latest WHO report reveals that men, including those who identify as gay and bisexual, constitute most mpox cases in Kenya and Uganda. The two countries have recorded their first cases, and has put queer rights organizations and health care centers that treat the LGBTQ community on high alert.
The Uganda Minority Shelters Consortium, for example, confirmed to the Washington Blade that the collaboration with health service providers to prevent the spread of mpox among gay and bisexual men is “nascent and uneven.”
“While some community-led health service providers such as Ark Wellness Clinic, Children of the Sun Clinic, Ice Breakers Uganda Clinic, and Happy Family Youth Clinic, have demonstrated commendable efforts, widespread collaboration on mpox prevention remains a significant gap,” UMSC Coordinator John Grace stated. “This is particularly evident when compared to the response to the previous Red Eyes outbreak within the LGBT community.”
Grace noted that as of Wednesday, there were no known queer-friendly health service providers to offer mpox vaccinations to men who have sex with men. He called for health care centers to provide inclusive services and a more coordinated approach.
Although Grace pointed out the fear of discrimination — and particularly Uganda’s Anti-Homosexuality Act — remains a big barrier to mpox prevention through testing, vaccination, and treatment among queer people, he confirmed no mpox cases have been reported among the LGBTQ community.
Uganda so far has reported two mpox cases — refugees who had travelled from Congo.
“We are for the most part encouraging safer sex practices even after potential future vaccinations are conducted as it can also be spread through bodily fluids like saliva and sweat,” Grace said.
Grace also noted that raising awareness about mpox among the queer community and seeking treatment when infected remains a challenge due to the historical and ongoing homophobic stigma and that more comprehensive and reliable advocacy is needed. He said Grindr and other digital platforms have been crucial in raising awareness.
The declarations of mpox as a global health emergency have already attracted demand for global leaders to support African countries to swiftly obtain the necessary vaccines and diagnostics.
“History shows we must act quickly and decisively when a public health emergency strikes. The current Mpox outbreak in Africa is one such emergency,” said ONE Global Health Senior Policy Director Jenny Ottenhoff.
ONE is a global, nonpartisan organization that advocates for the investments needed to create economic opportunities and healthier lives in Africa.
Ottenhoff warned failure to support the African countries with medical supplies needed to tackle mpox would leave the continent defenseless against the virus.
To ensure that African countries are adequately supported, ONE wants governments and pharmaceutical companies to urgently increase the provision of mpox vaccines so that the most affected African countries have affordable access to them. It also notes 10 million vaccine doses are currently needed to control the mpox outbreak in Africa, yet the continent has only 200,000 doses.
The Blade has reached out to Ishtar MSM, a community-based healthcare center in Nairobi, Kenya, that offers to service to gay and bisexual men, about their response to the mpox outbreak.
Health
White House urged to expand PrEP coverage for injectable form
HIV/AIDS service organizations made call on Wednesday
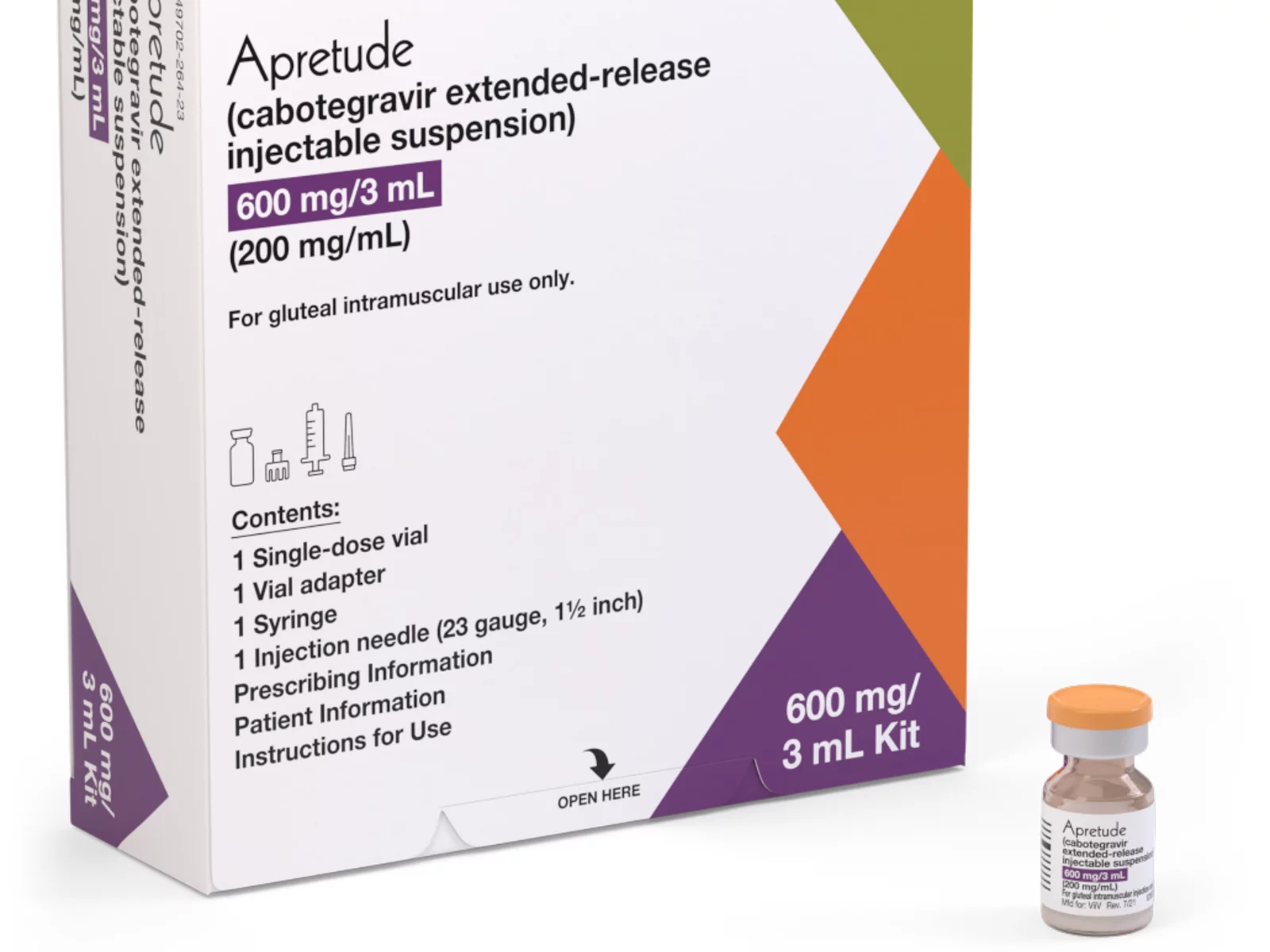
A coalition of 63 organizations dedicated to ending HIV called on the Biden-Harris administration on Wednesday to require insurers to cover long-acting pre-exposure prophylaxis (PrEP) without cost-sharing.
In a letter to Chiquita Brooks-LaSure, administrator of the Centers for Medicare and Medicaid Services, the groups emphasized the need for broad and equitable access to PrEP free of insurance barriers.
Long-acting PrEP is an injectable form of PrEP that’s effective over a long period of time. The FDA approved Apretude (cabotegravir extended-release injectable suspension) as the first and only long-acting injectable PrEP in late 2021. It’s intended for adults and adolescents weighing at least 77 lbs. who are at risk for HIV through sex.
The U.S. Preventive Services Task Force updated its recommendation for PrEP on Aug. 22, 2023, to include new medications such as the first long-acting PrEP drug. The coalition wants CMS to issue guidance requiring insurers to cover all forms of PrEP, including current and future FDA-approved drugs.
“Long-acting PrEP can be the answer to low PrEP uptake, particularly in communities not using PrEP today,” said Carl Schmid, executive director of the HIV+Hepatitis Policy Institute. “The Biden administration has an opportunity to ensure that people with private insurance can access PrEP now and into the future, free of any cost-sharing, with properly worded guidance to insurers.”
Currently, only 36 percent of those who could benefit from PrEP are using it. Significant disparities exist among racial and ethnic groups. Black people constitute 39 percent of new HIV diagnoses but only 14 percent of PrEP users, while Latinos represent 31 percent of new diagnoses but only 18 percent of PrEP users. In contrast, white people represent 24 percent of HIV diagnoses but 64 percent of PrEP users.
The groups also want CMS to prohibit insurers from employing prior authorization for PrEP, citing it as a significant barrier to access. Several states, including New York and California, already prohibit prior authorization for PrEP.
Modeling conducted for HIV+Hep, based on clinical trials of a once every 2-month injection, suggests that 87 percent more HIV cases would be averted compared to daily oral PrEP, with $4.25 billion in averted healthcare costs over 10 years.
Despite guidance issued to insurers in July 2021, PrEP users continue to report being charged cost-sharing for both the drug and ancillary services. A recent review of claims data found that 36 percent of PrEP users were charged for their drugs, and even 31 percent of those using generic PrEP faced cost-sharing.
The coalition’s letter follows a more detailed communication sent by HIV+Hepatitis Policy Institute to the Biden administration on July 2.
Signatories to the community letter include Advocates for Youth, AIDS United, Equality California, Fenway Health, Human Rights Campaign, and the National Coalition of STD Directors, among others.
-

 U.S. Supreme Court3 days ago
U.S. Supreme Court3 days agoSupreme Court upholds ACA rule that makes PrEP, other preventative care free
-

 U.S. Supreme Court3 days ago
U.S. Supreme Court3 days agoSupreme Court rules parents must have option to opt children out of LGBTQ-specific lessons
-

 National5 days ago
National5 days agoEvan Wolfson on the 10-year legacy of marriage equality
-
 Congress4 days ago
Congress4 days agoSenate parliamentarian orders removal of gender-affirming care ban from GOP reconciliation bill










