Local
Local gay student blocked from making blood donation despite FDA easing policy
Red Cross acknowledges delays, study to examine further change


A gay D.C. native otherwise qualified to donate blood was denied that chance at a local hospital, demonstrating that obstacles remain after the FDA eased its ban on gay donations — even as the agency has set up a pilot study to assess lifting the blanket policy altogether.
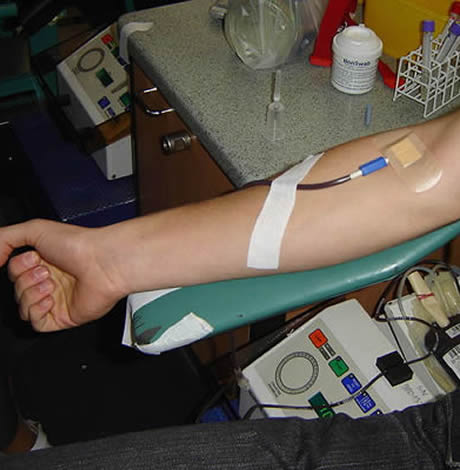
Aubrey Lay, a 19-year-old student at the College of William & Mary, told the Washington Blade after the FDA eased its policy last month he registered with his family at Children’s National Hospital, a D.C.-based pediatrics center, in hopes of making a contribution under the belief he’d meet the new requirements.
“As a gay man, I was very excited that the FDA had relaxed its guideline restrictions on MSM donating blood, however imperfect this change may be,” Lay said. “This meant that for the first time since graduating high school, I would be able to donate blood.”
Lay said he would have been ineligible to donate under the former rule requiring 12 months of abstinence for gay men, but believed he would have been eligible under the new rules requiring only three months. Although the hospital’s website had the old information online, Lay said he proceeded to his appointment on April 29 under the “wishful thinking” the hospital would accept him.
But Lay was ultimately blocked from making a donation.
“It turned out I was giving them the benefit of the doubt where I shouldn’t have,” Lay said. “I signed in and had my intake interview during which I explained my sexual history, and that I had last had sexual contact in early January, over the three-month limit. After answering all these questions, I got my finger pricked and iron levels checked, and height, weight, [blood pressure] taken. Only then was I told that I would not be able to donate because the hospital’s policy has not yet caught up to national standards.”
Lay said he was “shaken” by the experience, having wanted to make a blood donation to help out as the nation battles the coronavirus crisis.
As evidence going to the hospital was a risk he didn’t take lightly, Lay said it was the first time he’d left his house in a month because he and his family took the Virginia stay-at-home order “very seriously.”
“I certainly would not have risked my own health, and that of others, had I known I would be turned away,” Lay said.
Amid the blood shortage during the coronavirus pandemic, the FDA last month announced it would ease the ban on blood donations for various groups, including men who have sex with men.
The earlier ban, established in 2015, required gay men to be abstinent for 12 months before making a donation, but the new policy eased those restrictions, requiring a deferral period of three months.
Before 2015, the FDA had a lifetime ban on blood donations from men who have had sex with men. The policy was implemented in 1983 at the height of the AIDS epidemic based on fears donations from gay men would allow HIV to enter the blood supply.
Lay, upon being denied the ability to donate blood, said he found the office of the hospital’s president next to the blood donation room and sought an audience.
The president, Lay said, apologized for the confusion and connected him with the doctor in charge of the blood bank. The doctor, Lay said, told him the hospital’s computer systems were interlaced with the new policy and — in normal times without coronavirus — it would take a month to disentangle them and make them consistent with the new policy.
The website for the Children’s National Hospital has been updated and no longer indicates it follows the earlier 12-month policy, but is in the process of making a transition to conform to the new guidance by June 2020.
A Children’s National Hospital spokesperson confirmed the center is still in the process of updating its policy for gay donors and expects resolution next month.
“We have the deepest appreciation for all our blood donors and see every day how their donation saves lives,” the spokesperson said. “We take every step to adhere to all regulations around blood donation and are working to update our policies, donor questionnaire and computer systems to align to the new FDA regulations. The FDA just approved the donor questionnaire for sites to use and we are hoping to have all policies in place in June. We encourage anyone who would like to donate to make an appointment and double check on the eligibility before coming in.”
Lay’s story isn’t unique as other gay men have reported being unable to donate blood after the FDA eased its ban. Whether or not each of these men expected to meet the requirements under the new deferral period of three months is unclear.
Gay TV personality Andy Cohen says he was denied the chance to donate, even though his plasma would be rich in the antibody for COVID-19. Lukus Estok, a young gay man who recovered from COVID-19, also told his story about being denied the opportunity to make a donation at the New York Blood Center. His Facebook post went viral on social media and was featured in a Good Morning America article.
Mathew Lasky, a spokesperson for the LGBTQ media watchdog GLAAD, which has been leading calls for a change in blood policy, said more and more stories are emerging of LGBTQ people being blocked from making donations despite the recent change.
“GLAAD is continuing to pressure the FDA to put science above stigma, and to remove any time-based deferral on gay and bisexual men, and others in the LGBTQ community, donating blood,” Lasky said. “In the meantime, it’s critical for individual blood centers to begin adhering to the new FDA guidelines as not to continue needlessly discriminating against LGBTQ people, and to address the critical blood supply shortages we’re seeing during COVID-19.”
The American Red Cross, which had called for the FDA to shorten its deferral period from the 12 months to three months before the new policy was implemented, cautioned via a spokesperson the implementation of updated rules “will take time.”
“As the largest blood provider in the country, we are working to implement the updated FDA eligibility changes and will provide an update on timeline in the near future,” the spokesperson said. “It’s important to note that the implementation process includes potentially thousands of individuals and involves complex system updates that do take time. We are working to develop a reinstatement process to share with potential donors in the near future.”
UPDATE: A Red Cross spokesperson after publication of this article told the Blade a June 1 target date has been set for full implementation of the FDA policy change.
“The American Red Cross is working to implement the updated FDA eligibility changes on June 1,” the spokesperson said. “We just want to emphasize that this process includes potentially thousands of individuals and involves system updates that will take time to implement.”
Even as blood centers work to update their system to conform to the new policy, LGBTQ advocates have said the change is insufficient and the FDA should scrap its deferral requirements entirely for men who have sex with men and instead implement a policy based on individual risk assessment. Such a policy could enable, for example, monogamous gay men or gay men with safe sex practices to make blood donations.
Among those calling for this change is California Attorney General Xavier Becerra, who last month led a letter to the Department of Health & Human Services with 19 other attorneys general urging the Trump administration to abandon the deferral period, citing the success of other countries, such as Spain, Italy and Portugal, that have implemented gender-neutral policies.
Many voices have emerged calling for any and all obstacles to gay men making blood donations being lifted because testing could determine whether a blood donation has HIV. Experts, however, say some caution is necessary as a window exists between donation and testing in which the test wouldn’t be able to determine if a donation was free of the virus.
Scott Schoettes, counsel and the HIV Project Director at Lambda Legal, articulated that warning in a conference call last month with reporters.
“People believe that it catches all new cases of HIV or new cases of Hepatitis B, Hepatitis C, other blood borne infections,” Schoettes said. “That is not the case. There is still a window period during which that testing would not uncover a newly acquired infection.”
But in a rare situation of hitting both the gas and brake at the same time amid a policy change, the FDA has indicated the agency also is in the early stages of a pilot program to determine whether or not the ban can be changed further in favor of a risk-based assessment.
Megan McSeveney, an FDA spokesperson, confirmed the agency is set to begin the study with “about 2,000 men who have sex with men and who would be willing to donate blood.”
“This study, being conducted at community health centers in key locations across the United States, could generate data that will help the FDA determine if a donor questionnaire based on individual risk assessment would be as effective as time-based deferrals in reducing the risk of HIV,” McSeveney said.
The Red Cross is participating in the efforts to ensure a new risk-based policy as opposed to a deferral period will keep the blood supply safe, a spokesperson for the non-profit said.
“We are very early in the study – we are currently finalizing the content of the new questions, and developing the specifics of the protocols, including site selection, which then will require government and ethical review board approval,” the spokesperson said. “The results of testing in combination with the responses to the questionnaire will be used to determine the value of the questions in interdicting new HIV infections.”
Based on the limited information of the study, there’s no indication a change will, in fact, follow eliminating the deferral period for gay men entirely in favor of a risk-based assessment.
Sarah Kate Ellis, CEO of GLAAD, said in a statement the pilot study was wholly necessary given medical experts who say any kind of deferral period is “stigma, not science.”
“During the current crisis, the FDA is wasting time and money on a pilot study when all the scientific research and medical authorities plainly state that gay and bi man should not be restricted from donating blood,” Ellis said. “All blood donations, regardless of sexual orientation, are screened to ensure healthy samples and now the American Medical Association, leading elected officials, and more than 600 medical professionals have all done the work for the FDA and unequivocally state that this ban needs to end.”
Lay, caught in the middle of reconsideration of the policy, said he awaits the day when the restrictions are changed so LGBTQ people can finally donate blood at a time when their help is needed.
“Because of this crisis, blood donation centers should not be turning people away at the door who are by all measures qualified donors, risking their health and the health of those around them by their very presence in these spaces,” Lay said. “It is so important that we make this discrepancy between policy and practice known so that we can push for more transparency in blood donation, and hopefully full implementation of the new policy, albeit imperfect.”
Maryland
‘Girlfriends’ wanted for murder in Silver Spring, Md.
Montgomery County police say two charged with killing mother of one of them

The Montgomery County, Md., Department of Police announced on June 4 that it is seeking the public’s help in locating two women, who they identify as a couple, who are charged with first-degree murder for allegedly killing the mother of one of them.
In a statement police identified the two women as Vanessa Tjongarero-Henderson of Clarksburg, Md., and her girlfriend Samantha Raebel of Phoenix, Ariz. The statement says the two are charged with the murder of Hilde Henderson, 67, the mother of Vanessa.
According to the statement, officers with the department’s 3rd District found Hilde Henderson deceased on Tuesday, May 26, 2026, at her home at the Charter House apartments in the 1300 block of Fenwick Lane in Silver Spring after being called to check on the resident’s welfare.
“Henderson was transported to the Office of the Chief Medical Examiner, where an autopsy was conducted,” the statement says. “The cause of death was ruled a homicide.”
It adds, “Through the course of the investigation, detectives identified Henderson’s daughter, Vanessa Tjongarero-Henderson and Vanessa’s girlfriend, Raeble, as the suspects.” It says detectives obtained an arrest warrant against the two women, charging both with first-degree murder.
“Anyone with information regarding the location of these suspects or this crime is asked to call 911 or to visit the Crime Solvers of Montgomery County, Md. website at crimesolversmcmd.org,” the statement says, or to call the tip line at 1-866-411-8477.
“Tips with information leading to an arrest may be eligible for a reward from $250 up to $10,000,” it says, adding that tips may remain anonymous.
A spokesperson for Montgomery County police didn’t immediately respond to a request from the Washington Blade for information not disclosed in the police statement, including the physical-medical cause of death for Hilde Henderson and whether detectives have determined a motive for the murder.
District of Columbia
D.C. Latinx Pride celebrates culture and heritage
Your guide to events throughout June

Organizers with the Latinx History Project have planned a host of events this Pride season with parties, poetry, drag and more.
The festivities begin with the DC Latinx Pride 2026 Kickoff at Crush Dance Bar (2007 14th Street, N.W.) on Friday, June 12 from 6-10 p.m. The party will include a coronation ceremony for the 2026 Royal Court: Ms. DC Latinx Pride Vida Rangel and Mx. DC Latinx Pride Steph Niaupari. RSVP at latinxhistoryproject.org. The event is free, though donations are accepted.
An outdoor event is planned for Sunday, June 14 from 11 a.m.-1 p.m. at Anacostia River Park (1500 Anacostia Dr., S.E.). Cultivating Queer Outdoor Joy is a “peaceful outdoor community event focused on grounding, connection, and queer joy in nature.” The event is free.
A panel discussion is planned for The Festival Center (1640 Columbia Rd., N.W., 2nd floor) on Monday, June 15 from 6-8 p.m. La Plática: The Future of 2 Spirits and Trans Natives will focus upon the “stories, leadership and vision of Two-Spirit, Indigiqueer and Trans Native people.” RSVP to the free event at latinxhistoryproject.org.
A sex-positive poetry workshop, “Hoetry: Writing Erotic Poetry,” is planned for Wednesday, June 17 from 6-8 p.m. at The Festival Center (1640 Columbia Road, N.W.). The event is free.
The workshop So You Wanna Do Drag? is planned for Thursday, June 18 from 5:30-8 p.m. at The Festival Center (1640 Columbia Road, N.W.). Featured guests Ricky Rose and Mari Con Carne will hold a style showcase to discuss the basics of developing a drag persona. RSVP to the free event at latinxhistoryproject.org.
The Latinx History Project is collaborating with Rumba Queer DC to produce an official Latinx Pride Party: Sin Vergüenza. The event is at the multi-level venue, Transmission (1353 H Street, N.E.) on Thursday, June 18 from 7 p.m.-1 a.m. There are dance lessons, vendors and three different music experiences in the sprawling venue. There will also be a drag showcase from 10-11 p.m. The event is 21+ and tickets are available at shotgun.live/en/events/sin-verguenza. Tickets are $15 for entry into the party. Tickets to participate in the dance lesson are $29.98. Participants may choose between a bachata lesson or a salsa lesson from 7-8 p.m.
La Fiesta: Official DC Latinx Pride Party is planned for Friday, June 19 from 10 p.m. – 2 a.m. at Bunker (2001 14th Street, N.W.). Serena Morena from “Drag Race México” and “Drag Race UK vs The World” is slated to headline the 21+ event. Early tickets are available for $15 (plus $0.38 service fee) until June 16. The door cover charge without early tickets is $20. Attendees can also purchase a meet and greet experience with Serena Morena for $30. Tickets are available at latinxhistoryproject.org.
The Latinx History Project plans to march in the Capital Pride Parade on Saturday, June 20 and to have a table at the Capital Pride Festival on Sunday, June 21. Visit latinxhistoryproject.org to register to march alongside LGP in the parade or to staff the table at the festival.
The DC Latinx Pride 2026 Closing Event is scheduled for Friday, June 26 from 6:30-8:30 p.m. at the Mexican Cultural Institute (2829 16th Street, N.W.). The free event is a panel discussion “centering the experiences of immigrants who have lived in Latin America and now call the United States home.”
Visit latinxhistoryproject.org for more information.
District of Columbia
JR.’s hosts meet & greet for mayoral candidate Janeese Lewis George
Event organized by Capital Stonewall Democrats, Queers for Janeese

D.C. mayoral candidate Janeese Lewis George spoke to a crowd of LGBTQ supporters on June 1 at a meet & greet event held at JR.’s on 17th Street in the Dupont Circle neighborhood.
The event, organized by Capital Stonewall Democrats, which has endorsed Lewis George for mayor, with support from a group called Queers for Janeese, was followed by a “get out the vote” canvassing endeavor in which several of those attending the meet & greet visited the homes of nearby residents known to be Lewis George supporters.
The purpose of the canvassing was to remind Lewis George supporters to return their mail-in ballots or go to the polls on June 16 to elect Lewis George as the city’s next mayor, according to Matthew Kavanagh, one of the leaders of Queers for Janeese who attended the meet & greet event at JR.’s.
Local political observers consider Lewis George, a Ward 4 D.C. Council member, and former At-Large D.C. Council member Kenyan McDuffie, to be the two leading candidates in this year’s race for mayor. The two are among seven mayoral candidates competing in the city’s June 16 Democratic primary.
Lewis George told those attending the meet & greet, which was held on the JR.’s outdoor patio, that she has a long record of advocating for and initiating city polices and laws in support of the LGBTQ community. She said large corporate donors were backing her opponents and urged her LGBTQ supporters to help raise funds for her in the remaining days of the campaign.
Among those attending the meet & greet was gay longtime Dupont Circle civic activist Randy Downs who last November opened a nearby eatery called Protest Pizza. “I am queer and I am a Janeese supporter,” Downs told the Blade.
Stevie McCarty, president of Capital Stonewall Democrats, who also spoke at the meet & greet event, said his group would organize events in support of Lewis George in the remaining days of the campaign. Among them, he said, was an LGBTQ bar crawl in which supporters of Lewis George, including the candidate herself, would visit LGBTQ bars to promote her candidacy.

-

 Celebrity News5 days ago
Celebrity News5 days agoOutright International honors Cyndi Lauper at annual NYC gala
-

 Health5 days ago
Health5 days agoAIDS Healthcare Foundation announces 3 million people globally in its care
-

 National5 days ago
National5 days agoResults from key Tuesday primary races
-

 U.S. Military/Pentagon4 days ago
U.S. Military/Pentagon4 days agoFederal appeals court rules White House illegally banned trans troops