National
HIV research sped development of COVID vaccine
Top NIH official says success in coronavirus will boost AIDS work

Since 1996, Carl W. Dieffenbach, who holds a Ph.D. in biophysics from John Hopkins University, has served as director of the Division of AIDS at the National Institute of Allergies and Infectious Diseases, which is an arm of the U.S. National Institutes of Health or NIH.
In a June 10 interview with the Washington Blade, Dieffenbach gave an update on the extensive, ongoing research into the development of an HIV/AIDS vaccine that he has helped to coordinate for many years, including current human trials for a prospective AIDS vaccine taking place in the U.S., South America, and Africa.
One thing he feels passionate about is a development not widely reported in the media reports about the successful development of the COVID-19 vaccine. According to Dieffenbach, the extensive research into an AIDS vaccine in recent and past years, while not yet successful in yielding an effective AIDS vaccine, helped lay the groundwork for the rapid development of the different versions of a COVID vaccine.
“Because my division runs the largest clinical trials program in the word, we jumped in with both feet to help with coronavirus disease for both vaccines and drugs and things like that,” he said. “And the platforms that were used – the way they are making the coronavirus vaccines – the RNA vaccines with Moderna – were first piloted by NIH and Moderna to try to make an HIV vaccine,” Dieffenbach says.
“So, in many ways, the work for the past 25 years that we’ve done in HIV vaccines sped the development of coronavirus vaccines,” he told the Blade. “And now it’s time to take what we’ve learned from coronavirus and take it back to HIV and start afresh or continue with what we have and build upon from what we have learned.”
Dieffenbach says one reason the development of a COVID vaccine came about before an AIDS vaccine, despite more than 20 years of AIDS vaccine research, is that the HIV virus is far more complex than the coronavirus, especially its ability to infect and remain embedded in the infected person for life.
“Back in 2007 we had the first hint that an AIDS vaccine might be possible with a study called RV144,” Dieffenbach says. “We spent 10 years trying to replicate that, and we just completed that study – a study called HVTN702. And it showed no efficacy,” he said, meaning it did not work.
“So that was a big disappointment to us,” he says “But in the meantime, we had pushed forward with the J&J [Johnson and Johnson pharmaceutical company] vaccine and are pretty far along. We’ll see what happens. We should know in the next several months whether the N26 version of an AIDS vaccine, and HIV vaccine works or not,” he says. “We’re very close to an answer.”
Washington Blade: Where do things stand in the development of an HIV/AIDS vaccine in light of Dr. Fauci’s statement a few weeks ago that the development of a COVID-19 vaccine could provide a boost to developing an AIDS vaccine?
Carl Dieffenbach: Sure. So, maybe I can start by introducing myself to you as a way of putting this into a context.
So, I’m the director of the Division of AIDS, which is the largest funder of HIV research in the world. And I report directly to Dr. Fauci. So, I’m responsible for all AIDS, all the time. And that is my passion and purpose in life. Part of that is working toward a safe, effective, and durable HIV vaccine, which has been one of the two most challenging questions left in science today. The other is a cure. They are connected in some ways.
So, with that as background, when coronavirus disease came along – because my division runs the largest clinical trials program in the world – we jumped in with both feet to help with coronavirus disease for both vaccines and drugs and things like that. And the platforms that were used – the way they are making the coronavirus vaccines – the RNA vaccines with Moderna were first piloted by NIH and Moderna to try to make an HIV vaccine. So, we’ve being working on that platform with Moderna for several years.
The leadership at Pfizer used to be part of a group at Penn, where we were also working with them. The J&J vaccine – we currently have in two Phase III clinical trials for HIV, one in sub-Saharan Africa, specifically in young women and the other one in the Americas in men who have sex with men and transgender individuals. Both of those Phase IIIs are moving along. The women’s study is fully enrolled. The men’s study was hit hard by COVID, but we worked through and will be fully enrolled by September.
One other vaccine just to talk about is the Oxford vaccine, the AstraZeneca vaccine. That is also using a platform at Oxford University, which has been used for HIV. So, in many ways, the work for the past 25 years that we’ve done in HIV vaccines sped the development of coronavirus vaccines. And now it’s time to take what we’ve learned from coronavirus and take it back to HIV and start afresh or continue with what we have and build upon from where we have learned.
Blade: That’s very interesting. But can we assume, then, from the clinical trials that have taken place for an HIV vaccine that they did not succeed in providing the immunity needed for an effective vaccine?
Dieffenbach: So, that’s exactly the problem we have. Back in 2007 we had the first hint that an AIDS vaccine might be possible with a study called RV144. We spent 10 years trying to replicate that, and we just completed that study – a study called HVTN702. And it showed no efficacy. So, that was a big disappointment to us. But in the meantime, we had pushed forward with the J&J vaccine and are pretty far along. We’ll see what happens. We should know in the next several months whether the N26 version of an AIDS vaccine, and HIV vaccine works or not. We’re very close to an answer.
Blade: So, the human trials are ongoing.
Dieffenbach: Oh, again – the study in young women in sub-Sahara Africa is fully enrolled. The men’s study will be fully enrolled in September. So, we have fought through the coronavirus epidemic to maintain, to nurse these trials along to make sure with the $100 million or so we’ve invested, that we didn’t want them to go down the drain literally because we lost too many people for follow-up. So, this was a herculean effort that has gone on all the time trying to do the vaccine studies for coronavirus disease, which we were also incredibly successful in.
Blade: Can we assume all of the people participating in the studies were HIV negative?
Dieffenbach: Yes, they’re HIV negative. They are people who are at risk. And also, in South America, for example, the major countries we’re in are Peru and Brazil. And they’ve had a strong research culture with us, going back more than a decade. For example, both of those countries played big roles in our studies of pre-exposure prophylaxis. A study called I-PREX that demonstrated that in men who have sex with men that [a PrEP drug] works well to prevent HIV acquisition in seronegative men who have sex with men.
So, we’ve been there. This is a really good setup for the countries, for the citizens that are in those countries that want to avail themselves to the research that has benefited everybody.
Blade: Among those who are participating in these ongoing AIDS vaccine trials, can we assume they cannot be taking the PrEP anti-retroviral drugs that have been shown to be highly effective in preventing HIV infection?
Dieffenbach: So, what we’ve done is we – everything is by conversation. So, when somebody who is interested in the study comes in, we talk to them. What is your chief interest in being in this study? And a lot of people want to be in the study because then they can access PrEP. They want to make it easier to get a hold of pre-exposure prophylaxis. They feel that is the best way that they can protect themselves.
So, in that situation, what we do is we take those people and link them to PrEP services where they can easily get PrEP in their community. So, first it’s taking care of those people. Then there are people who really have no interest in PrEP. And we actually counsel them every time they come in for a study. Are you sure you don’t want to access PrEP? And those are the people we then say, if you’re not interested in PrEP, what do you think about participating in a vaccine trial?
Because they’re the ones who have the most freedom of thought. They don’t have an opinion about the vaccine or about PrEP. So, those are the people we’ve been focusing on and enrolling. So, we’ve been very careful to make sure that if people wanted PrEP they not only have access, but they didn’t feel like somehow having to trade something in order to get it. The freedom to join a study should be a free choice. And it shouldn’t be a coercive thing to get PrEP. So, we just took that off the table and said if you’re truly interested in PrEP we can get you PrEP and make sure that was available.
Blade: So, in that case, if they choose PrEP they would not be in the vaccine trial?
Dieffenbach: You know, it’s interesting that you ask it in that way. Because you have relationships with your community, many of the investigators have reported that people will say, you know I tried PrEP and it wasn’t for me. It made me gaseous. It upset my stomach. I wasn’t myself. I tried it. I couldn’t make it work for me. I want to stop PrEP. Am I still eligible for the [vaccine] study? And the answer is of course. Many people are very happy on PrEP and they come in for visits occasionally and say this is working for me and just have the relationship with the doctors there, so it works. So, again, it’s about maintaining contact with your communities.
Blade: Can you tell a little about what happens next after people become part of an HIV vaccine trial. Do you have to keep in touch with these people, and do they have to get an HIV test periodically?
Dieffenbach: Exactly. So, the vaccine consists of a series of injections. It’s a mixture of vector systems that delivers a series of encoded HIV genes that are specifically designed to induce very broad immunity. There’s a whole computer-based process to design those components of the vaccine to make sure that it has sequence similarities with all the different versions of HIV circulating in the globe. And then at the end there is a protein boost. And we carry this out.
So, about every three to four months people come in. They get a shot. They fill out questionnaires. They give a blood sample. And they’re tested for HIV and are given a boost or a placebo. And they stay in touch with the clinic. They come in and out of the clinic. And the retention is quite high in these situations because people really like having the attention of the clinic available to them. It’s part of the community.
Blade: So, they go to a clinic for all of this?
Dieffenbach: It’s a research clinic. It’s not like a state-run health clinic. It’s a research clinic. Clinic is just a term for where people are seen.
Blade: Are any of these AIDS vaccine trials that are going on taking place in the United States?
Dieffenbach: Yes. So, the study is called Mosaico. And it’s HVTN706. And we have sites throughout the United States as well as South America. But that study is limited to men who have sex with men – the one in the United States.
Blade: Is it broader than just men who have sex with men in other countries?
Dieffenbach: No, so we decided to really focus on specific at-risk populations. So, in the Americas we chose to focus on men who have sex with men and transgender individuals. And sub-Saharan Africa we focused on young women because that is the target of the study population. So, 705 is all women in sub-Saharan Africa. And in the Americas in North and South America it is all men who have sex with men and transgender individuals.
Blade: Can we assume that the researchers that are doing these studies have a sensitivity of LGBTQ people? Is there still an issue where people worry about being outed as being gay or transgender?
Dieffenbach: So, many of the sites that we work with have been part of our system for over 20 years. And so, they are trusted members of the LGBTQ community within their cities and states. And ‘states’ is a literal term where it’s a state in Colombia or Peru or Brazil. And so, it is part of the fabric of the gay community in these places. Just like in San Francisco the San Francisco health clinic and the DCF clinics are part and parcel of everything the community does there.
And so, the lead physician in San Francisco is Susan Buchbinder. She has been a leader in health in this population for over 25 years or actually closer to 30 years at this point. We’re all getting old. Do you know that? So, we have been at this a very long time. And really have tried to build structures that are durable and therefore are reliable to the community. And that’s where we go back to the same groups time after time.
Blade: Have the locations of the vaccine testing sites been released publicly?
Dieffenbach: Yes, all of that is publicly available on clinicaltrials.gov. If you go into clinicaltrials.gov and search HVTN705 or HVTN706 you will get a version of the protocol, all the times it’s been modified, where we are – the protocol. All of that is public knowledge and available to you. HVTN705 is the women’s study. HVTN706 is the men’s study.
Blade: Is there a timeframe for when these latest vaccine studies might be completed?
Dieffenbach: I think within the next several months. We will get an answer out of the women’s study and then the men’s study is probably a year away. We were slowed a little bit because of COVID. We actually had to pause enrollment for several months. But we’re back on track.
Blade: Isn’t there a parallel research effort for an HIV/AIDS cure?
Dieffenbach: Yes, we have a very large program in cure research. It is a lot earlier in the discovery process and so it’s still very ‘researchy.’ And we have a very large program called the Martin Delany Collaboratories for Cure Research. Martin Delany was an activist who really pushed NIH in so many wonderful ways to really take the need for a cure seriously. His argument was a cure is the next logical step after effective anti-retroviral therapy. You cannot stop with one pill once a day. You’ve got to keep going. And he was pretty persistent. And unfortunately, he died several years go and we just thought the best way to honor him, and his memory was to name a program after him.
Editor’s note: Next week, in the second and final installment of his interview with the Blade, Dr. Dieffenbach discusses the progress in research and studies into an HIV/AIDS cure and explains from a scientific standpoint why an HIV vaccine is taking longer to develop than a COVID vaccine.
National
Madonna turns Times Square into massive dance floor
Pop icon celebrates Pride month with surprise performance

Pop icon Madonna celebrated Pride month with a pop-up performance in New York City’s Times Square on Thursday to the delight of 50,000 fans.
She performed for about 15 minutes high above street level, including several songs from her new album “Confessions II” due on July 3, along with a trio of songs from the first “Confessions on a Dance Floor.”
In addition to the brand new “Love Sensation,” she performed “I Feel So Free” and “Bring Your Love,” plus “Hung Up,” “Get Together” and “I Love New York.” She wished the crowd a happy Pride season; the event was shared with audiences through Grindr’s first-ever livestream.


National
Gallup finds LGBTQ support among Americans is dropping
Marriage equality support lowest since 2016

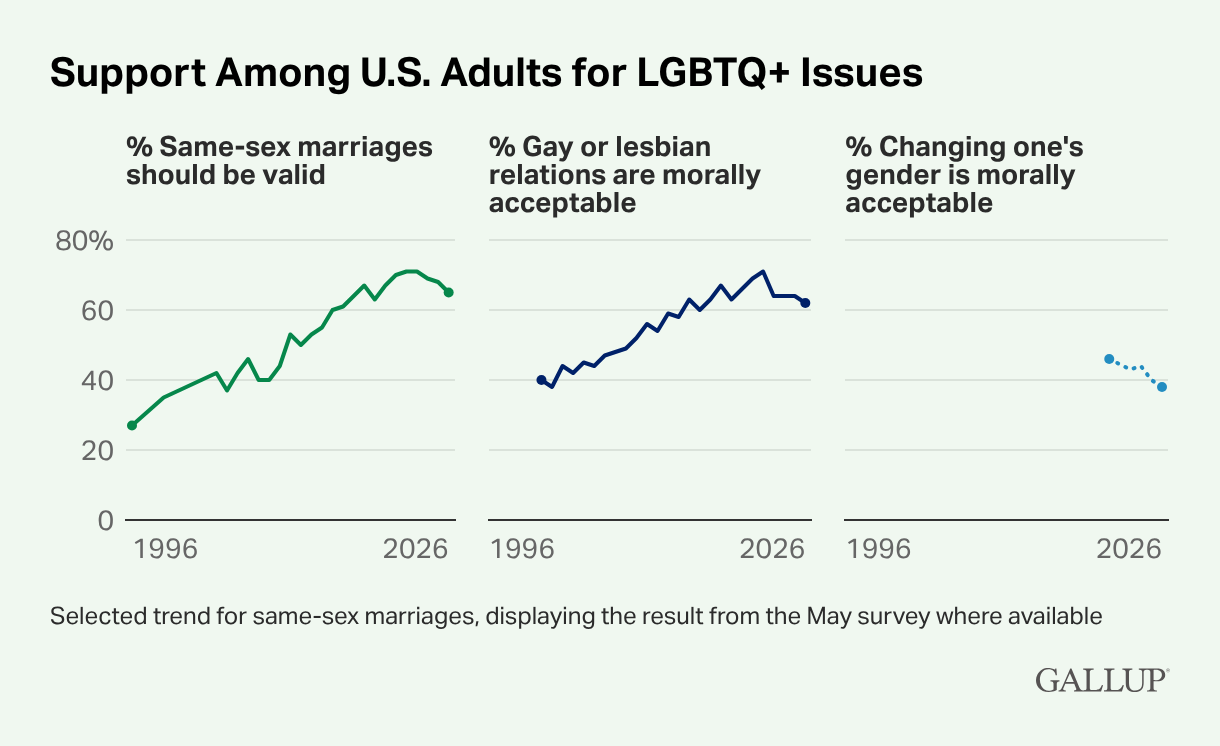
Gallup, one of the leading organizations in public opinion polling, has found that LGBTQ support among Americans is dropping.
The poll, whose data was collected using Gallup’s annual Values and Beliefs survey, was conducted in May and was published on Wednesday. The data was collected through telephone interviews from a sample of more than 1,000 adults living in all 50 states and D.C. using random digit dialing.
It highlights declining attitudes surrounding LGBTQ issues in multiple areas — from support for same-sex marriage to views on gender identity and the morality of one’s sexuality.
One of the most striking findings was that support for marriage equality fell six points from its 2022-2023 high.
The survey also found that 62 percent of Americans view gay and lesbian relations as morally acceptable, the lowest level since 2016 just after same-sex marriage was legalized nationwide by the U.S. Supreme Court.
One newer question on the poll found that the perceived morality of changing one’s gender has dropped eight points since 2021, indicating the American public is less supportive of transgender people.
The data attributes much of the decline to shifting Republican views alongside the party itself. Conservative leaders have pushed back against diversity, equity, and inclusion programs that were intended to foster greater acceptance of LGBTQ people and other historically disadvantaged groups.
President Donald Trump has been a guiding force behind waves of anti-LGBTQ sentiment, particularly when it comes to trans rights. The president has enacted multiple executive orders, including Executive Order 14168, “Defending Women from Gender Ideology Extremism and Restoring Biological Truth to the Federal Government,” which mandates that gender be defined by one’s sex assigned at birth. He also signed Executive Order 14183, “Prioritizing Military Excellence and Readiness,” which barred qualified trans applicants from joining the military and led to the removal of trans service members already serving in the armed forces.
Additionally, he signed Executive Order 14201, “Keeping Men Out of Women’s Sports,” which prohibits trans female athletes from participating on women’s and girls’ sports teams.
In February, Gallup found that an estimated 9 percent of Americans identified as part of the LGBTQ community in some form.
The organization also found that 23 percent of adults under age 30 identify as LGBTQ, compared with 10 percent of those ages 30 to 49 and 3 percent or less among those ages 50 and older.
Congress
Ogles faces bipartisan backlash over anti-gay social media post
Tenn. congressman blamed the comment on staffer

U.S. Rep. Andy Ogles (R-Tenn.), who represents Tennessee’s 5th Congressional District, is facing backlash from LGBTQ advocates and fellow Republicans after a social media post declared that “homosexuality has no place in America.”
“Homosexuality has no place in America. Happy Nuclear Family Month,” the congressman wrote in a post on X that was later deleted.
According to the Williams Institute at UCLA School of Law, an estimated 6.3 percent of U.S. adults identify as LGBTQ.
Following widespread criticism, Ogles removed the post and blamed it on a staff member.
“The post was stupid, hurtful and a complete distraction from my America First focus. The employee has been reprimanded,” Ogles said in a statement.
The Washington Blade reached out to Ogles’s office for comment but did not receive a response by press time.
Among those condemning the message was U.S. Rep. Mike Lawler (R-N.Y.), who called it “absolutely idiotic” in a social media post.
“Homosexuality exists. In America,” Lawler wrote on X. “In fact, Andy, you have family, friends, neighbors, colleagues, and constituents who are gay and lesbian. It doesn’t make them less than or somehow unworthy of being an American.”
U.S. Sen. Ted Cruz (R-Texas) also criticized Ogles’s remarks.
“For all of recorded history, homosexuals have been a part of humanity,” Cruz told TMZ DC. “I think the behavior of consenting adults is their business.”
Chris Sanders, the executive director for the Tennessee Equality Project and Tennessee Equality Project Foundation provided a statement to the Blade about Ogles’s comment.
“The Tennessee Nuclear Family Month resolution has really backfired on conservatives by ensnaring Congressman Ogles in scandal. He used the resolution as a pretext to say that our community doesn’t belong in America, resulting in incredible backlash from across the partisan divide,” Sanders said. “It is a good opportunity for him to pause and reflect on whether it’s time for him to resign. Fighting one’s own constituents is not the purpose of serving in Congress.”
Human Rights Campaign Senior Press Secretary Jarred Keller provided a statement to the Blade regarding Ogles’s comments.
“LGBTQ+ people are woven into the fabric of America, and any politician who questions that is severely out of touch with reality. When so many people are worried about whether they can afford gas to get to work or groceries for their families, the last thing we need is right-wing Republicans targeting marginalized communities with hateful attacks,” Keller said. “Representative Ogles should spend less time attacking LGBTQ+ people and start addressing the issues that actually matter, because last I checked, our community isn’t the reason families are struggling to make ends meet.”
The controversy comes as Tennessee continues to advance legislation affecting LGBTQ residents. The state already has several laws on the books that LGBTQ advocates have criticized, including the Adult Entertainment Act, enacted in 2023, which restricts certain “adult cabaret performances.”
Lawmakers have also introduced additional measures this legislative session, including the “No Pride Flag or Month Act,” which would prohibit state employees, volunteers, and agents from displaying Pride flags or participating in Pride observances while acting in an official capacity.
Another proposal, the “Banning Bostock Act” would seek to limit the application of state anti-discrimination protections based on the U.S. Supreme Court’s decision in Bostock v. Clayton County. Tennessee lawmakers have also passed other measures restricting LGBTQ rights and access to gender-affirming health care.



















