Local
Local gay student blocked from making blood donation despite FDA easing policy
Red Cross acknowledges delays, study to examine further change


A gay D.C. native otherwise qualified to donate blood was denied that chance at a local hospital, demonstrating that obstacles remain after the FDA eased its ban on gay donations — even as the agency has set up a pilot study to assess lifting the blanket policy altogether.
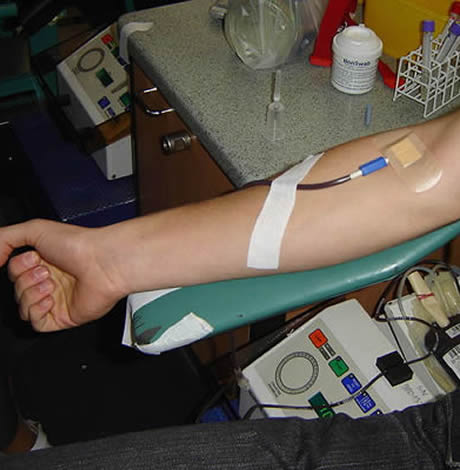
Aubrey Lay, a 19-year-old student at the College of William & Mary, told the Washington Blade after the FDA eased its policy last month he registered with his family at Children’s National Hospital, a D.C.-based pediatrics center, in hopes of making a contribution under the belief he’d meet the new requirements.
“As a gay man, I was very excited that the FDA had relaxed its guideline restrictions on MSM donating blood, however imperfect this change may be,” Lay said. “This meant that for the first time since graduating high school, I would be able to donate blood.”
Lay said he would have been ineligible to donate under the former rule requiring 12 months of abstinence for gay men, but believed he would have been eligible under the new rules requiring only three months. Although the hospital’s website had the old information online, Lay said he proceeded to his appointment on April 29 under the “wishful thinking” the hospital would accept him.
But Lay was ultimately blocked from making a donation.
“It turned out I was giving them the benefit of the doubt where I shouldn’t have,” Lay said. “I signed in and had my intake interview during which I explained my sexual history, and that I had last had sexual contact in early January, over the three-month limit. After answering all these questions, I got my finger pricked and iron levels checked, and height, weight, [blood pressure] taken. Only then was I told that I would not be able to donate because the hospital’s policy has not yet caught up to national standards.”
Lay said he was “shaken” by the experience, having wanted to make a blood donation to help out as the nation battles the coronavirus crisis.
As evidence going to the hospital was a risk he didn’t take lightly, Lay said it was the first time he’d left his house in a month because he and his family took the Virginia stay-at-home order “very seriously.”
“I certainly would not have risked my own health, and that of others, had I known I would be turned away,” Lay said.
Amid the blood shortage during the coronavirus pandemic, the FDA last month announced it would ease the ban on blood donations for various groups, including men who have sex with men.
The earlier ban, established in 2015, required gay men to be abstinent for 12 months before making a donation, but the new policy eased those restrictions, requiring a deferral period of three months.
Before 2015, the FDA had a lifetime ban on blood donations from men who have had sex with men. The policy was implemented in 1983 at the height of the AIDS epidemic based on fears donations from gay men would allow HIV to enter the blood supply.
Lay, upon being denied the ability to donate blood, said he found the office of the hospital’s president next to the blood donation room and sought an audience.
The president, Lay said, apologized for the confusion and connected him with the doctor in charge of the blood bank. The doctor, Lay said, told him the hospital’s computer systems were interlaced with the new policy and — in normal times without coronavirus — it would take a month to disentangle them and make them consistent with the new policy.
The website for the Children’s National Hospital has been updated and no longer indicates it follows the earlier 12-month policy, but is in the process of making a transition to conform to the new guidance by June 2020.
A Children’s National Hospital spokesperson confirmed the center is still in the process of updating its policy for gay donors and expects resolution next month.
“We have the deepest appreciation for all our blood donors and see every day how their donation saves lives,” the spokesperson said. “We take every step to adhere to all regulations around blood donation and are working to update our policies, donor questionnaire and computer systems to align to the new FDA regulations. The FDA just approved the donor questionnaire for sites to use and we are hoping to have all policies in place in June. We encourage anyone who would like to donate to make an appointment and double check on the eligibility before coming in.”
Lay’s story isn’t unique as other gay men have reported being unable to donate blood after the FDA eased its ban. Whether or not each of these men expected to meet the requirements under the new deferral period of three months is unclear.
Gay TV personality Andy Cohen says he was denied the chance to donate, even though his plasma would be rich in the antibody for COVID-19. Lukus Estok, a young gay man who recovered from COVID-19, also told his story about being denied the opportunity to make a donation at the New York Blood Center. His Facebook post went viral on social media and was featured in a Good Morning America article.
Mathew Lasky, a spokesperson for the LGBTQ media watchdog GLAAD, which has been leading calls for a change in blood policy, said more and more stories are emerging of LGBTQ people being blocked from making donations despite the recent change.
“GLAAD is continuing to pressure the FDA to put science above stigma, and to remove any time-based deferral on gay and bisexual men, and others in the LGBTQ community, donating blood,” Lasky said. “In the meantime, it’s critical for individual blood centers to begin adhering to the new FDA guidelines as not to continue needlessly discriminating against LGBTQ people, and to address the critical blood supply shortages we’re seeing during COVID-19.”
The American Red Cross, which had called for the FDA to shorten its deferral period from the 12 months to three months before the new policy was implemented, cautioned via a spokesperson the implementation of updated rules “will take time.”
“As the largest blood provider in the country, we are working to implement the updated FDA eligibility changes and will provide an update on timeline in the near future,” the spokesperson said. “It’s important to note that the implementation process includes potentially thousands of individuals and involves complex system updates that do take time. We are working to develop a reinstatement process to share with potential donors in the near future.”
UPDATE: A Red Cross spokesperson after publication of this article told the Blade a June 1 target date has been set for full implementation of the FDA policy change.
“The American Red Cross is working to implement the updated FDA eligibility changes on June 1,” the spokesperson said. “We just want to emphasize that this process includes potentially thousands of individuals and involves system updates that will take time to implement.”
Even as blood centers work to update their system to conform to the new policy, LGBTQ advocates have said the change is insufficient and the FDA should scrap its deferral requirements entirely for men who have sex with men and instead implement a policy based on individual risk assessment. Such a policy could enable, for example, monogamous gay men or gay men with safe sex practices to make blood donations.
Among those calling for this change is California Attorney General Xavier Becerra, who last month led a letter to the Department of Health & Human Services with 19 other attorneys general urging the Trump administration to abandon the deferral period, citing the success of other countries, such as Spain, Italy and Portugal, that have implemented gender-neutral policies.
Many voices have emerged calling for any and all obstacles to gay men making blood donations being lifted because testing could determine whether a blood donation has HIV. Experts, however, say some caution is necessary as a window exists between donation and testing in which the test wouldn’t be able to determine if a donation was free of the virus.
Scott Schoettes, counsel and the HIV Project Director at Lambda Legal, articulated that warning in a conference call last month with reporters.
“People believe that it catches all new cases of HIV or new cases of Hepatitis B, Hepatitis C, other blood borne infections,” Schoettes said. “That is not the case. There is still a window period during which that testing would not uncover a newly acquired infection.”
But in a rare situation of hitting both the gas and brake at the same time amid a policy change, the FDA has indicated the agency also is in the early stages of a pilot program to determine whether or not the ban can be changed further in favor of a risk-based assessment.
Megan McSeveney, an FDA spokesperson, confirmed the agency is set to begin the study with “about 2,000 men who have sex with men and who would be willing to donate blood.”
“This study, being conducted at community health centers in key locations across the United States, could generate data that will help the FDA determine if a donor questionnaire based on individual risk assessment would be as effective as time-based deferrals in reducing the risk of HIV,” McSeveney said.
The Red Cross is participating in the efforts to ensure a new risk-based policy as opposed to a deferral period will keep the blood supply safe, a spokesperson for the non-profit said.
“We are very early in the study – we are currently finalizing the content of the new questions, and developing the specifics of the protocols, including site selection, which then will require government and ethical review board approval,” the spokesperson said. “The results of testing in combination with the responses to the questionnaire will be used to determine the value of the questions in interdicting new HIV infections.”
Based on the limited information of the study, there’s no indication a change will, in fact, follow eliminating the deferral period for gay men entirely in favor of a risk-based assessment.
Sarah Kate Ellis, CEO of GLAAD, said in a statement the pilot study was wholly necessary given medical experts who say any kind of deferral period is “stigma, not science.”
“During the current crisis, the FDA is wasting time and money on a pilot study when all the scientific research and medical authorities plainly state that gay and bi man should not be restricted from donating blood,” Ellis said. “All blood donations, regardless of sexual orientation, are screened to ensure healthy samples and now the American Medical Association, leading elected officials, and more than 600 medical professionals have all done the work for the FDA and unequivocally state that this ban needs to end.”
Lay, caught in the middle of reconsideration of the policy, said he awaits the day when the restrictions are changed so LGBTQ people can finally donate blood at a time when their help is needed.
“Because of this crisis, blood donation centers should not be turning people away at the door who are by all measures qualified donors, risking their health and the health of those around them by their very presence in these spaces,” Lay said. “It is so important that we make this discrepancy between policy and practice known so that we can push for more transparency in blood donation, and hopefully full implementation of the new policy, albeit imperfect.”
Maryland
Silver Spring holds annual Pride In The Plaza
‘Today means inclusion. It means to build resilience’

Silver Spring’s annual Pride in the Plaza event took place on Sunday to celebrate the LGBTQ community and emphasize inclusion and resilience.
“Today means inclusion. It means to build resilience, love,” Robyn Woods, program and outreach director for Live In Your Truth, which organized the event, said. “I mean, just being surrounded by the community and so many great entrepreneurs, business owners, and just being a part of this whole rainbow coalition that we call the LGBTQIA to be about.”
With the event being her first time organizing for Live In Your Truth, Woods said she felt emotional to see the support and love at the event.
“Some people (are) bringing out their children, their babies, their grandparents,” Woods said. “It’s a lot more allies here than anything else. That type of support to me means so much more than just support from my community; just outside support, inside support, so much support around it, so much love. Everyone’s smiling outside, helping each other.”
Attendees of the event were able to head over to the Family Fun Zone, an air-conditioned Pride Cool Down Lounge, or watch live drag performances in the main stage area.
Along with entertainment and a shaved-ice stand, rows of information tables stood along the plaza, including FreeState Justice, the Washington Spirit, Trans Maryland, Moco Pride Center, and the Heartwood Program, an organization that offers support, therapy, education, and resources to the LGBTQ community.
“I want people to know about our services, and I love what we have to offer,” Jessica Simon, psychotherapist for Heartwood Program’s Gender Wellness Clinic, said. “I (also) want to be part of a celebration with the community, and so it feels good to be here with other people who have something they want to give to the community.”
She added that within today’s political climate, to which she called an “antidote to shame,” it’s important to be celebrating Pride.
“There’s a lot of demonization of LGBTQI people,” Siena Iacuvazzi, facilitator for Maryland Trans Unity, said. “(Pride) is part of the healing process.”
Iacuvazzi said she was taught to be ashamed of who she was growing up, but being a part of a community helped her flourish in the future.
“I was taught how to hate myself. I was taught that I was an abomination to God,” she said. “But being a community is like understanding that there are people who have experienced the same thing, and they’re flourishing. They’re flourishing because they’re willing to stand up for themselves as human beings and discover themselves and understand what’s true for themselves.”
She added that Pride allows for a mutual understanding to take place.
“It’s more of a sense of belonging … and just taking that home and understanding you’re not alone,” Iacuvazzi said. “We’re each taking our own journey — we’re not putting that on each other. It’s just walking away with a sense of belonging and humanity.”
Similar to Iacuvazzi, Woods said she hopes attendees’ biggest takeaways would be family, fun, resilience, and pride.
“Being proud of yourself, being happy for who you are, and representation and how much it matters,” she continued. “And I think all these young people that are walking around here get to see versions of themselves, but older. They get to see so many different lesbian, gay, bisexual, pansexual people that are successful, that are showing love, that care, and it’s not how we’re portrayed in the media. It’s lovely to see it out here. (It’s) like we’re one big old, happy family.”
Virginia
Spanberger touts equality, reproductive rights in Arlington
Democratic Va. gubernatorial nominee made campaign stop at Freddie’s Beach Bar

With the general election heating up and LGBTQ rights under increasing threat nationwide, Virginia gubernatorial candidate Abigail Spanberger brought her “Span Virginia Bus Tour” to Arlington’s Freddie’s Beach Bar for a campaign stop filled with cheers, policy pledges, and community spirit.
Spanberger, who served three terms in the U.S. House of Representatives from 2019 through early 2025 for Virginia’s 7th Congressional District, also served as a federal law enforcement officer specializing in narcotics and money laundering cases, and as a CIA case officer working on counterterrorism and nuclear counterproliferation.
Spanberger is running against Republican nominee Winsome Earle-Sears, the current lieutenant governor of Virginia, who said she was “morally opposed” to a bill protecting marriage equality in the commonwealth.
She was joined by other Democratic candidates and supporters: lieutenant gubernatorial candidate Ghazala Hashmi, attorney general candidate Jay Jones, Virginia state Sen. Adam Ebbin (D-Alexandria), and Congressman Don Beyer.

Freddie’s was packed wall-to-wall with supporters, many of whom wore “Spanberger for Virginia” shirts in the progressive Pride flag colors. In her speech, she made it clear that LGBTQ Virginians’ rights are on the ballot this year.
“I’m so excited to be here, and I am so grateful to the entire staff of Freddy’s for letting us overtake this incredible venue that is not just an awesome place to come together in community, but is a symbol to so many people of joy, of happiness, of community and of celebrating our friends and our neighbors,” Spanberger told the packed restaurant. “It is exciting to be here, and particularly during this Pride month, and particularly as we reflect on the 10-year anniversary of Obergefell and the reality that we still have so much work to do.”
“The reality is there are so many people who still would be inclined to take us backwards,” she said. “In this moment when we see attacks on people’s rights, on people’s humanity, on Virginia, on our economy, on research, on public education, on food security, on health care, on Virginians, on their jobs, on public service and on people — it can get heavy.”
“What it does for me is it makes me want to double down, because once upon a time, when I was talking to my mother about some horror show or sequence of activities coming out of a particular administration, she did not really have the patience to listen to me and said ‘Abigail, let your rage fuel you’ — and the conversation was over. And so I reflect on that, because, in fact, every day there is so much fuel to be had in this world and in this moment.”
One of the points Spanberger continued to emphasize was the importance of steadfast state government officials following the election of President Donald Trump, which has led to rollbacks of LGBTQ and bodily autonomy rights as a result of the conservative-majority U.S. Supreme Court.
“What the past few years have shown us is that a Supreme Court decision, no matter how many years we have celebrated its existence, does not protect us in the long term. And so as governor, I will work to make sure that every protection we can put in place for the dignity, the value, and the equal rights of all Virginians is a priority.”
During her speech, Spanberger highlighted several of the key values driving her campaign — protecting reproductive freedom and human rights, lowering healthcare costs, safeguarding Virginia’s environment, and ensuring that public education is affordable, accessible, and rooted in truth, not politics.
Spanberger went as far as to say that she wants to amend the state’s constitution to remove Section 15-A. “The reality is that in Virginia, we still have a ban in our state constitution on marriage equality. It is of the utmost urgency that we move forward with our constitutional amendment.”
“We will work to ensure that that terrible constitutional amendment, that was put in years ago, is taken out and updated and ensuring that Virginia is reflective in our most essential documents of who we are as a commonwealth, which is an accepting place that celebrates the vibrancy of every single person and recognizes that all Virginians have a place, both in that constitution and in law,” she added.
Following the event, two supporters spoke to the Washington Blade about why they had come out to support Spanberger.
“I came out because I needed to show support for this ticket, because it has been a particularly rough week, but a long few years for our rights in this country, in this state, with this governor, and it’s — we need to flip it around, because queer people need protection,” said Samantha Perez, who lives in Ballston. “Trans people need protection. Trans kids need protection. And it’s not gonna happen with who’s in Richmond right now, and we just need to get it turned around.”

“The whole neighborhood’s here. All our friends are here,” said Annie Styles of Pentagon City. “It means the world to me to take care of each other. That’s what a good community does. That’s not what we’ve had with the Republicans here or across the nation for a really long time. It’s time to show that care. It’s time to make sure that good people are in a position to do good things.”
District of Columbia
Activists protest outside Hungarian Embassy in DC
Budapest Pride scheduled to take place Saturday, despite ban

More than two dozen activists gathered in front of the Hungarian Embassy in D.C. on Friday to protest the country’s ban on Budapest Pride and other LGBTQ-specific events.
Amnesty International USA Executive Director Paul O’Brien read a letter that Dávid Vig, executive director of Amnesty International Hungary, wrote.
“For 30 years Budapest Pride has been a celebration of hope, courage, and love,” said Vig in the letter that O’Brien read. “Each march through the streets of Budapest has been a powerful testament to the resilience of those who dare to demand equality, but a new law threatens to erase Pride and silence everyone who demands equal rights for LGBTI people.”
“The Hungarian government’s relentless campaign against LGBTI rights represents a worrying trend that can spread normalizing division and hatred,” added Vig. “Thank you for standing with us when we refuse to be intimidated.”
Council for Global Equality Chair Mark Bromley and two of his colleagues — Stephen Leonelli and Keifer Buckingham — also spoke. Health GAP Executive Director Asia Russell and Chloe Schwenke, a political appointee in the Obama-Biden administration who worked for the U.S. Agency for International Development, and Planned Parenthood staffers are among those who attended the protest.
(Washington Blade video by Michael K. Lavers)
Hungarian lawmakers in March passed a bill that bans Pride events and allow authorities to use facial recognition technology to identify those who participate in them. MPs in April amended the Hungarian constitution to ban public LGBTQ events.
Budapest Pride is scheduled to take place on Saturday, despite the ban. Hundreds of European lawmakers are expected to participate.
“Sending strength to the patriotic Hungarians marching tomorrow to advance human dignity and fundamental rights in a country they love,” said David Pressman, the gay former U.S. Ambassador to Hungary, on Friday on social media.
Sending strength to the patriotic Hungarians marching tomorrow to advance human dignity and fundamental rights in a country they love. Szabadság és szerelem. My past remarks on Budapest Pride: https://t.co/y1QhA9QouA
— David Pressman (@AmbPressman) June 27, 2025
-

 U.S. Supreme Court3 days ago
U.S. Supreme Court3 days agoSupreme Court upholds ACA rule that makes PrEP, other preventative care free
-

 U.S. Supreme Court3 days ago
U.S. Supreme Court3 days agoSupreme Court rules parents must have option to opt children out of LGBTQ-specific lessons
-

 National5 days ago
National5 days agoEvan Wolfson on the 10-year legacy of marriage equality
-
 Congress4 days ago
Congress4 days agoSenate parliamentarian orders removal of gender-affirming care ban from GOP reconciliation bill