Coronavirus
NBC4 producer participates in coronavirus vaccine trial
Brandon Benavides is gay and Latino


Tens of thousands of people are participating in vaccine trials to bring an end to the coronavirus pandemic which has killed millions worldwide. One of these test subjects is Brandon Benavides, a gay, Latino writer and producer for NBC4.
Benavides said he heard that several different trials were looking for more Black and Latino people to participate and he wanted to help out. After being rejected from two trials, he was accepted into one conducted by Moderna, a Massachusetts-based Biotech firm.
“When I started telling people what I did, my friend told me, ‘Well Brandon, you’re helping humanity.’ And I didn’t think about that until she said it,” he told the Washington Blade during a recent interview. “I didn’t quite think about the impact this could have on the world.”
Moderna’s vaccine, dubbed mRNA-1273, is currently in phase three of its testing process and the company is preparing to distribute up to 20 million doses throughout the country by the end of the year.
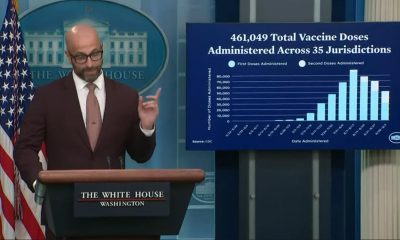
Chief Medical Officer Tal Zaks told investors in an Oct. 29 conference call that 37 percent of their 30,000 person testing pool comes from “diverse communities” and two thirds are over the age of 45.
“We’re ready,” Zaks said about distribution. “We expect MRNA-1273 to be distributed within existing infrastructure. There’s nothing new required that hasn’t already been used for years with many other vaccines.”
Benavides said he went through an extensive health screening before receiving his first dose in late September from the Optimal Research Clinic in Rockville, Md., although he isn’t sure whether the injection he received was the experimental vaccine. The trial is a double-blind study which means neither the subject nor the doctor administering the injection know if it’s the vaccine or a placebo.
Benavides said he even had to look away so he couldn’t see the labels on the vaccine.
Moderna’s vaccine comes in two installments, 28 days apart. Benavides said he only noticed side effects after the second injection, which he received on Oct. 2. He said his temperature rose by one degree 30 minutes after the shot and when he woke up for his midnight shift at NBC, he experienced cold sweats and a headache which he said is “not normal” for him.
Slightly concerned, he called the research clinic and received a call back the next day. The clinic told him that fatigue and the cold sweats were normal side effects of the vaccine.
“This is really where I felt like a guinea pig because all I could do was just tell them what was happening to me,” he said. “I don’t know if I have the vaccine or the placebo but I feel like I do have it because I felt that reaction after the second dose.”
Benavides said he has felt fine since that day.
Zaks told investors during last week’s conference call that the most common side effects of the vaccine are headache, fatigue, muscle pain, chills, and injection site pain which can be “mild to moderate in severity.” These symptoms are to be expected, Zaks said, and are caused when the vaccine prompts a response from the body’s immune system.
“Importantly, there were no vaccine-related serious adverse events in this trial and no patterns of concern for any clinical labs,” he said.
After getting the second dose, Benavides said he has started to slowly go out more often. He went to the grocery store for the first time in seven months and has restarted sessions with a personal trainer at the gym.
“Even though I may have this layer of protection, the vaccine, I’m cognizant of what’s going on,” he said. “There’s all these people and they’re all touching all these things and I know I need to keep my distance and take precautions.”
When reflecting on the study, Benavides said he will most likely never participate in a trial like this again.
“I was scared for the whole process. But I prepared myself, I knew what I was getting myself into,” he said. “I do feel like a science experiment but I also feel that this is bigger than me.”
Coronavirus
D.C. mayor to lift all restrictions on bars, nightclubs on June 11
‘We will definitely be celebrating Pride’ next month

D.C. Mayor Muriel Bowser announced at a news conference on Monday that a continuing trend of significantly lower numbers of coronavirus cases and deaths in the city has enabled her to fully lift capacity and other restrictions on most businesses, including restaurants and places of worship, on May 21.
The mayor said bars and nightclubs will be allowed to increase indoor capacity from the current 25 percent to 50 percent on May 21, with all capacity restrictions for bars and nightclubs to be removed on June 11.
The mayor’s announcement came after representatives of the city’s nightlife businesses, including the city’s gay bars and restaurants, expressed concern that D.C. had yet to lift its capacity restrictions beyond 25 percent while surrounding jurisdictions in Maryland and Virginia had already lifted most restrictions.
“On May 21, restrictions on public and commercial activity, including capacity limits, types of activities, and time restrictions, will be lifted,” the mayor’s directive says.
It says restrictions for bars and nightclubs would continue at a 50 percent capacity from May 21 through June 11. The directive says restrictions for large sports and entertainment venues would also continue from May 21 to June 11, which includes a requirement such events apply for a waiver of the restrictions on a case-by-case basis.
“On June 11, capacity limits and restrictions will be lifted on those venues that cannot fully reopen on May 21,” the directive says.
In response to a question at the news conference, Bowser said the June 11 date would essentially end all restrictions on nightclubs and bars, including the current requirement that they close at midnight rather than the pre-epidemic closing times of 2 a.m. on weekdays and 3 a.m. on weekends.
In a development that could have a major impact on plans for D.C.’s LGBTQ Pride events, the mayor’s revised health directive announced on Monday includes the lifting of all capacity restrictions on large outdoor and indoor sports and entertainment events beginning on June 11.
That change would remove restrictions that have, up until now, prevented D.C.’s Capital Pride Alliance from holding its annual Pride Parade and Festival in June during Pride Month.
Capital Pride Executive Director Ryan Bos told the Washington Blade shortly after the mayor’s announcement that Capital Pride is assessing its options for expanding its current plans for in-person events in June.
“We will definitely be celebrating Pride in June,” Bos said. “We just received this information as well. So, we will be getting further information,” he said. “We have not been informed that they will be issuing any permits yet, so at this time we are moving forward with our original plans for doing things.”
Bos was referring to a city requirement for obtaining permits for street closings and use of other public spaces for events such as a parade or street festival. He said existing plans, among other things, call for an informal parade of cars and other vehicles on June 12 that will drive throughout the city to view homes and businesses that will be decorated with Pride displays such as signs, photos, and other symbols of Pride.
Those familiar with the city’s past Pride events don’t think there will be enough time for Capital Pride to organize the traditional large parade and street festival in time for June. But Capital Pride officials have talked about holding a possible parade and festival in October, and the lifting of the capacity restrictions announced by Bowser on Monday would likely make that possible.
In addition to lifting all capacity restrictions on May 21 for restaurants, the mayor’s May 21 timeframe for lifting restrictions includes these additional venues and events:
- Weddings and special events
- Business meetings and seated conventions
- Places of worship
- Non-essential retail
- Personal services
- Private at-home gatherings
- Libraries, museums, galleries
- Recreation Centers
- Gyms and fitness centers
- Pools
- Office space
- Schools
- Childcare
“We’re very pleased that over the last several days, we have seen our case spread, our community spread numbers, venture out of the red into the yellow and fast approaching the green,” Bowser said in referring to a health department chart that shows the changes in coronavirus cases in the city.
“You might remember that our daily case rate peaked in January at 45.9. And today you can see it’s down to 6.6,” she said at her news conference on Monday.
“Throughout this process I have said how proud I am of D.C. residents and businesses who have responded, who have followed health guidance and have worked together to help protect our community throughout the pandemic. And we see it in these numbers today,” she said.
“Containing the virus will continue to require all of us to be focused on maintaining a robust health system,” the mayor said, adding that while over 200,000 D.C. residents have been fully vaccinated since December 2020, “many more thousands” still need to be vaccinated. “Vaccines are free and available on demand at walk-up sites across the District,” she said.
The mayor also noted that the city will continue to require residents and visitors to use a mask in accordance with existing and updated guidance set by the U.S. Centers for Disease Control and Prevention.
Mark Lee, coordinator of the D.C. Nightlife Council, an association that represents restaurants, bars, nightclubs and other entertainment venues, said the mayor’s directive on May 10 leaves some details to be addressed but will open the way to bring nightlife businesses back to life.
“What we do know is that on Friday, May 21, businesses begin returning to normal operations and, three weeks later, on June 11, all restrictions for all businesses in the District will end,” Lee said. “It’s a day we’ve long awaited and one that will save much of our community enterprise from financial ruin.”
Coronavirus
DC residents with HIV eligible for COVID vaccine
Mayor announces expanded eligibility as vaccine supply increases
D.C. Mayor Muriel Bowser announced on Feb. 24 that D.C. residents between the ages of 16 to 24 who have one of 19 pre-existing medical conditions, including HIV, will now be eligible to make an appointment to receive the COVID-19 vaccine.
The mayor and D.C. Department of Health Director Dr. LaQuandra Nesbitt said the appointments could be made through the city’s special site, vaccinate.dc.gov, beginning Thursday and Friday, Feb. 25 and 26. The vaccinations themselves for the expanded group of residents, including people with HIV, would begin March 1, the mayor said in an announcement.
Abby Fenton, a spokesperson for Whitman-Walker Health, the D.C. community health center that provides services to the LGBTQ community and people with HIV, said Whitman-Walker has begun contacting its HIV patients about the availability of the COVID vaccine for them.
“We are urging people to try to make an appointment with the city because we have such a limited supply,” Fenton said. She said Whitman-Walker is dispensing the vaccine for those who the city determines are eligible at its medical center locations at 1425 14th Street, N.W., and at its Max Robinson Center at 2301 Martin Luther King Jr. Ave., S.E.
Michael Kharfen, the DOH official in charge of the city’s HIV/AIDS, Hepatitis, STD, and Tuberculosis Administration, said the limited supplies of the COVID vaccine that the city has been receiving from the federal government has prevented the allocation of vaccine supplies to community health centers like Whitman-Walker until a few weeks ago.
He said supplies of the vaccine have increased in recent weeks and the Department of Health was hopeful that it will be able to provide additional supplies of the vaccine to community health centers and other facilities and health care providers.
Kharfen noted that the city has been increasing the availability of the vaccine to different groups of residents in stages as supplies have increased. Front line medical workers and nursing home residents were the first to receive the vaccine. The most recent group to become eligible prior to the mayor’s most recent expansion this week were people 65 years of age and older.
The mayor’s announcement on Feb. 24 listed these pre-existing medical conditions, including HIV, that would make city residents between the ages of 16 and 64 eligible for the COVID vaccine:
Asthma, Chronic Obstructive Pulmonary Disease (COPD), and other Chronic Lung Disease; Bone Marrow and Solid Organ Transplantation; Cancer; Cerebrovascular Disease; Chronic Kidney Disease; Congenital Heart Disease; Diabetes Mellitus; Heart Conditions, such as Heart Failure, Coronary Artery Disease, or Cardiomyopathies; HIV; Hypertension; Immunocompromised State; Inherited Metabolic Disorders; Intellectual and Developmental Disabilities; Liver Disease; Neurologic Conditions; Obesity, BMI ≥ 30 kg/m2; Pregnancy; Severe Genetic Disorders; Sickle Cell Disease; and Thalassemia.
Coronavirus
D.C. gay bars struggling to stay open in pandemic
Mayor’s new rule banning liquor sales after 10 p.m. called ‘devastating’


John Guggenmos, co-owner of the D.C. gay bars Number 9 and Trade, says he and his business partners support Mayor Muriel Bowser’s efforts to keep people safe as the number of people testing positive for COVID-19 continues to rise in the city.
But Guggenmos and other gay bar owners say the mayor’s most recent order requiring bars and restaurants to stop serving alcoholic beverages after 10 p.m. has had a devastating impact on what had already been a major decline in business since the COVID restrictions were put in place earlier this year.
“We see hope on the horizon,” Guggenmos said. “But for many places it’s just going to be too late. It is sad because even if I am in a position that we can weather this storm better, if other places in the neighborhood don’t, then we all suffer.”
David Perruzza, owner of the Adams Morgan gay sports bar Pitchers and its adjoining lesbian bar A League of Her Own, said gay bar customers traditionally come out to the clubs after 9 p.m. and often remain there several hours later.
Under the mayor’s current Phase II rules for addressing the COVID health emergency all restaurants and bars must close at midnight, two hours earlier than the pre-epidemic closing time of 2 a.m. during the week and three hours sooner than the normal 3 a.m. closing time on weekends. That restriction by itself has resulted in a significant drop in revenue for bars and nightclubs, including LGBTQ clubs, officials with the clubs have said.
The new restriction put in place last month banning liquor sales after 10 p.m. allows bars and restaurants to continue to stay open until midnight. But Guggenmos, Perruzza and other bar owners say few if any customers would likely come in to order non-alcoholic beverages. Thus they and nearly all of the city’s bar and restaurant owners have decided to close at 10 p.m. until the restrictions are lifted, a development that has further curtailed their businesses.
“I’ve had the worst two weekends of my life at the bar,” said Perruzza in referring to the weekends following the ban on liquor sales after 10 p.m. “I can’t sustain a business this way,” he said.

Dr. LaQuandra Nesbitt, director of the D.C. Department of Health, has said city inspectors have found that more violations of the COVID-related health restrictions at restaurants and bars, such as social distancing and mask wearing, were occurring after 10 p.m. as patrons consumed more alcohol. But nightlife advocates have disputed claims that riskier behavior occurs after 10 p.m. They say there are no studies or data to back up those claims.
Perruzza said he understands that while the mayor’s intention is to curtail the spread of the coronavirus he believes the 10 p.m. cutoff on alcohol service will result in large numbers of bar customers going to private parties in people’s homes where there will be fewer safeguards to curtail the virus.
“By her doing this she is going to push people to have more house parties,” Perruzza said. “At least if they’re in a restaurant or bar they’re in a controlled environment where they take their temperature. They make sure everything is sanitized after people leave,” Perruzza said. “People are not required to wear masks when they go to house parties.”
Prior to the start of the pandemic, D.C. was home to at least 15 gay bars or nightclubs in which the clientele was largely LGBTQ. A number of other D.C. bars and nightclubs are considered LGBTQ friendly, according to gay D.C. nightlife advocate Mark Lee, who said those additional establishments have a significant LGBTQ clientele.
In March, Bowser issued her initial emergency health order requiring all “non-essential” businesses, including bars and restaurants, to temporarily close their indoor operations to customers in an effort to curtail the spread of the coronavirus. Carryout food and drink orders were allowed, and some of the gay clubs joined other bars and restaurants in putting in place a take-out order business.
A short time later, the DC Eagle, the city’s longest continuously operating gay bar, announced it was permanently closing. The Eagle’s majority owner filed for Chapter 7 bankruptcy following longstanding financial problems, but many of the Eagle’s customers believe the pandemic played some role in the permanent shutdown.
At the same time, the popular LGBTQ nightclub Ziegfeld’s-Secrets closed its doors indefinitely after the owner of the building where it was located in the city’s Buzzard’s Point area invoked its longstanding plan to demolish the building to make way for a new condominium and retail development. Ziegfeld’s-Secrets manager Steve Delurba said the club would like to reopen in a new location but efforts to reopen would have to wait until all COVID-19 restrictions on such establishments were lifted.
Among the city’s remaining 13 LGBTQ bars and clubs, all but one has reopened after the mayor put in place the city’s Phase II business reopening plan in June, which allowed bars, restaurants, and other businesses to resume limited indoor operations.
The Fireplace, a gay bar at 2161 P St., N.W. near Dupont Circle, decided to remain closed rather than operate under the COVID restrictions but “definitely” plans to reopen, according Larry Ray, a longtime customer who said he spoke with one of the owners.
Among the other Phase II restrictions for bars, restaurants and nightclubs put in place by Bowser in the spring was the requirement that such establishments must operate at 50 percent of their normal indoor capacity, all patrons must be seated at tables spaced six feet apart, and at least three food items must be served that are prepared on the premises regardless of whether the establishment was exempt from serving food prior to the pandemic. The Phase II order also bans the establishments from offering live entertainment.
Two weeks ago, when the mayor issued her updated order banning the serving of alcoholic beverages after 10 p.m. at bars and restaurants, she also included in the order a reduction in the capacity of customers from 50 percent to 25 percent based on concern that the number of COVID-19 cases was rising in D.C. after the case number had gone down in the spring and summer.
Perruzza told the Blade that due to the Phase II social distancing requirements and the spacing of tables and the ban on allowing customers to stand except to walk in and out and go to the bathroom, Pitchers and his adjoining bar A League of Her Own were never able to reach a 50 percent capacity. At most, he said, he was able to reach a 33 percent capacity, which now must be reduced to 25 percent.
Meanwhile, the D.C. gay bar Dirty Goose at 913 U St., N.W. is among the establishments hit with a fine for allegedly violating the Phase II food serving requirement. According to a report in the Washington City Paper, an inspector from the city’s Alcoholic Beverage Regulation Administration on Nov. 27 cited Dirty Goose for serving only cookies as a food item, saying it failed to provide at least two other types of food such as popcorn or brownies instead of just cookies.
Co-owner Justin Parker told City Paper he plans to contest the citation on grounds that the establishment serves multiple types of cookies that are prepared on the premises and that the different types should be accepted as different food types. He said that ABRA inspectors came to Dirty Goose six or seven times in November prior to citing him for the food violation and found his establishment to be in full compliance with all of the COVID related requirements.
On its Facebook page the Dirty Goose announced on Nov. 10 that it had voluntarily closed its doors after one of its employees tested positive for COVID and out of caution it would remain closed until all remaining employees were tested. On Nov. 15 it announced “we have received all our employees test results and we are ready to reopen,” which happened less than a week later.
In a Nov. 25 Facebook message, Dirty Goose conveyed what appears to be the sentiment shared by the other LGBTQ bar owners and operators.
“First, we would like to thank all of our wonderful family of patrons who have kept us going since May,” the message says. “What a crazy 8 months it’s been!” After announcing the Dirty Goose would be closing at 10 p.m. each day due to the mayor’s order banning alcohol sales after that hour, the message added, “We look forward to continue serving all of you and please know we are continuously following the safety requirements set by the DOH and the safety of our staff and patrons remains our main priority.”

Lee, the longtime D.C. nightlife advocate who served as director of the D.C. Nightlife Council before being furloughed, said the 10 p.m. cutoff for the sale of liquor at bars and restaurants will be especially harmful coming with all the other restrictions.
“The most maddening aspect of Mayor Bowser suddenly halting all alcohol consumption after 10 p.m. at local bars, restaurants, and nightclubs operating in full compliance with public safety protocols and highly restricted service limitations is that there is no actual data or evidence-based rationale for this financially devastating roll-back curfew,” Lee told the Blade.
“This arbitrary edict jeopardizes the survival of hospitality establishments by causing them to lose the major portion of revenue they had been able to generate,” he said. “We’re getting reports that this decision is costing operators up to 60 percent or more of the little money they were making, leaving most with no choice other than to shut down two hours earlier rather than attempt to now serve only food items and non-alcoholic beverages until midnight,” Lee said.
Lee noted that at a press conference on Dec. 7, Bowser acknowledged that nightlife establishments, including restaurants and bars, have done an exemplary job of complying with health requirements and providing a safe space for patrons and employees.
At that press conference the mayor also said she looks forward to being able to lift all restrictions on businesses once the COVID vaccine becomes widely available. But she said that with a resurgence of COVID cases in D.C. in recent weeks along with the rise in cases in the surrounding suburbs the city could be forced once again to order the complete shutdown of indoor operations of businesses like restaurants and bars if the local COVID situation worsens.
Perruzza, Guggenmos and Doug Schantz, owner of the gay sports bar Nellie’s at 900 U St., N.W., each said their establishments and others like them serve as a place where LGBTQ people can go to be themselves, which many are unable to do at work, school, or even at home in some situations.
“At some point safe human interactions are what people are craving,” said Guggenmos. “You see someone on the street and how they desperately just want that interaction again,” he said. “If we could do that safely, why not?”
D.C.’s LGBTQ Bars/Restaurants
Nellie’s Sports Bar
900 U Street, N.W.
202-332-6355
Uproar
639 Florida Ave., N.W.
202-462-4464
The Dirty Goose
913 U Street, N.W.
JR.’s
1519 17th Street, N.W.
202-328-0090
Windows/DIK Bar
Upper floor at Dupont Italian Kitchen
1637 17th Street, N.W.
202-328-0100
Annie’s Paramount Steakhouse restaurant/bar
1609 17th Street, N.W.
202-232-0395
Larry’s Lounge
1840 18th Street, N.W.
202-483-1483
Pitchers/League of Her Own
2317 18th Street, N.W.
202-733-2558
Duplex Diner
2004 18th Street, N.W.
202-265-7828
The Fireplace
2161 P Street, N.W.
202-293-1293
[Temporarily closed during pandemic]
Number Nine
1435 P Street, N.W.
202-986-0999
Trade
1410 14th Street, N.W.
202-986-1094
Green Lantern
1335 Green Court, N.W.
202-347-4533
D.C. LGBTQ-friendly Bars/Clubs
Dacha Beer Garden
1600 7th Street, N.W.
202-350-9888
9:30 Club
815 V Street, N.W.
202-265-0930
DC 9 Nightclub
1940 9th Street, N.W.
202-483-5000
Dito’s Bar
Lower floor at Floriana Restaurant
1602 17th Street, N.W.
202-667-5937

-

 2026 Midterm Elections4 days ago
2026 Midterm Elections4 days agoBree Fram’s congressional campaign ends but her fight continues
-

 Celebrity News4 days ago
Celebrity News4 days agoPeppermint made her mark on ‘Drag Race.’ Now, her advocacy is front and center
-

 a&e features4 days ago
a&e features4 days agoFrom Media Matters to massive queer ragers: the rise of Tara Dikhof
-

 Opinions4 days ago
Opinions4 days agoWhy this Black Pride, I ranked Janeese Lewis George #1 for D.C. mayor